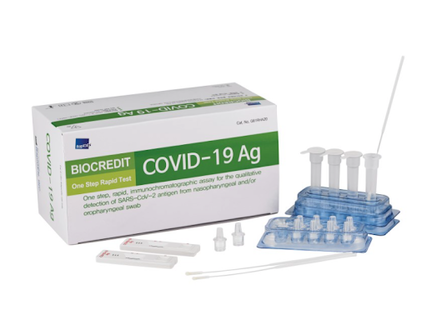
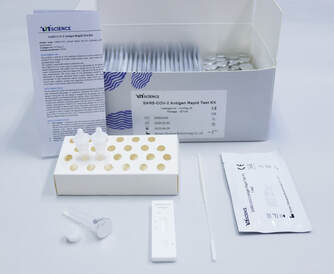
Biocredit COVID 19 Ag Rapid TestAsk for Quotation: eu@genprice.com or us@genprice.com
Size: 20 Tests/Kit Price: 50 In Stock |
|
Features
- The first commercialization Black Gold Particle technology In the world (high sensitivity and specificity) - User friendly Dual Color System (Control line: Red, Test line: Black) - Intended Use: Detection of SARS-CoV2 Antigen - Package: 20 Tests/Kit - Storage: 1~40℃ - Specimen Type: Nasopharynx /Nasopharyngeal - Shelf life: 12 months from manufacture date - Time to result: 5~8 minute |
|
COVID-19 Overview
2019 novel coronavirus (2019-nCoV) is a single-stranded RNA coronavirus. Coronavirus disease 2019 (COVID-19) is a respiratory illness caused by the 2019-nCoV belongs to the Beta-coronavirus Genus, which also includes Severe Acute Respiratory Syndrome coronavirus (SARS-CoV, 2003) and Middle East Respiratory Syndrome coronavirus (MERS-CoV, 2012). Coronaviruses, 2019-nCoV consist of four viral proteins named spike (S), envelope (E), Membrane (M), and nucleocapsid (N), Common signs of infection include respiratory syndromes, fever, cough, shortness of breath and breathing difficulties. In more severe cases, infection can cause pneumonia, severe acute respiratory syndrome, kidney failure and even death. General recommendations to prevent infection spread include regular hand washing, covering mouth and nose when coughing and sneezing. Avoid close contact with anyone experiencing symptoms of respiratory illness such as coughing and sneezing.
Principal of Test
BIOCREDIT COVID-19 Ag is a rapid chromatographic immunoassay for the qualitative detection of SARS-CoV-2 antigen in human nasopharynx. this test is for in-vitro professional diagnostic use and intended as an aid top early diagnosis of SARS-CoV-2 infection in patient with clinical symptoms. It provides only an initial screening test result and more specific alternative diagnosis methods should be performed in order to confirm COVID-19 infection,
Kit Components
- Each test device sealed in a foil pouch with a desiccant
- Assay diluent tube
- Filter cap
- Sterilized swab for nasopharynx specimen collection
- Instructions for use
Specimen Collection and Storage
- Specimen should be handled carefully as an infectious agent and should be collected by trained personnel.
- As improper collection of the sample affects the test result significantly, handle with care.
- More accurate results can be obtained if samples are collected from several parts.
- Specimen should be tested as soon as possible upon collection, if the sample has to be stores, store the swab sample at 2 - 8°C up to 4 hours prior to testing.
To collect nasopharyngeal swab specimen, tilt the patient's head slightly backwards, insert a nasopharyngeal swab horizontally into the nasal cavity until resistance is met at the level of the turbinate. Rotate the swab gently against the nasopharyngeal mucosa for 10 - 15 seconds. remove the swab while making sure that the tip of the swab is wet.
*When collect the specimens, follow the Instruction for use thoroughly.
Assay Procedure
[Preparation]
- Equilibrate kit components and specimen to room before testing.
- Do not break the seal of the foil pouch until ready to perform the test.
[Testing]
- Remove the aluminium seal from the assay diluent tube, Immerse nasopharyngeal swab in the assay diluent and swirl the swab 5 - 10 times while pressing the head against the bottom and side of the collection tube.
- Withdraw the swab while pinching and squeezing against the tube. Dispose it with biosafety.
- Close the assay diluent tube with a filter cap securely.
- Remove the device from the foil pouch and place it on a flat and dry surface.
- Invert the assay diluent tube and gently squeeze it to draw 3 - 4 drops (90 - 150 uL) into a sample well (S) of the device. *Please ensure that an appropriate amount of specimen and assay diluent is used for testing. Too much or too little amount of specimen and /or assay diluent may lead to deviation of results.
- Read the result between 5 - 8 minutes.
Interpretation of Results
[Negative]
The presence of only one red brand at the control line (C) within result window indicates a negative resukt.
[Positive]
Two brands appear, one red control line (C) and one black test line (T).
[Invalid]
If the control line fails to appear within the result window, the result is considered invalid. The directions may not have been followed correctly or the test may have deteriorated. It is recommended that the specimen be retested.
Note: There is no meaning attributed to line color intensity or width.
The presence of only one red brand at the control line (C) within result window indicates a negative resukt.
[Positive]
Two brands appear, one red control line (C) and one black test line (T).
[Invalid]
If the control line fails to appear within the result window, the result is considered invalid. The directions may not have been followed correctly or the test may have deteriorated. It is recommended that the specimen be retested.
Note: There is no meaning attributed to line color intensity or width.